Relationship between Orthochromatic dye and Metachromatic dye
-
In chemistry Orthochromatic refers to a dye or stain which does not change colour on binding to a target as opposed to Metachromatic stains which change colour. In lateral sense it comes from ortho i.e straight or correct chromatic i.e coloured. As for example Toluidine colour but it stains mast cell granules by becoming Metachromatic which in this case is red.
-
Metachromatic is a characteristic change in the colour of staining carried out in biological tissues, exhibited by certain aniline dyes when they bind to particular substances present in these tissues, chronotropes. For example Toluidine blue becomes pink when bound to cartilage. The absence of colour change in staining is named orthochromatic.
-
In spectral terms Orthochromatic refers to maintaining the position of the spectral peaks and metachromatic in contract refers to a shift in the position which can be either hypsochromic or bathochromic depending on it’s direction.
-
The underlying mechanism for metachromasia is the presence of polyanions within the tissue. When these tissues are stained with a concentrated basic dye solution, such as toluidine blue, the dye molecules are close enough to form dimeric and polymeric aggregates. The absorption properties of these aggregations differ from those of the individual nonaggregated dye molecules.
-
Orthochromatic photography refers to a photographic emulsion that is sensitive to only blue and green light, and thus can be processed with a red safelight. The increased blue sensitivity causes blue objects to appear lighter and red ones darker. A cyan lens filter which removes red light, can be used with standard panchromatic film produce a similar effect.
Toluidine Blue O (7-amino-8-methylphenothiazin-3-ylidene)-dimethylammonium chloride
[ C15H16N3S•Cl]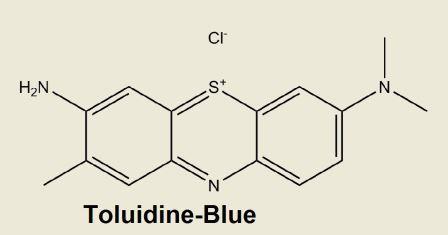
Erythrosine (Red No. 3) IUPAC name 2-(6-Hydroxy-2,4,5,7-tetraiodo-3-oxo-xanthen-9-yl) benzoic acid
[C20H6I4Na2O5]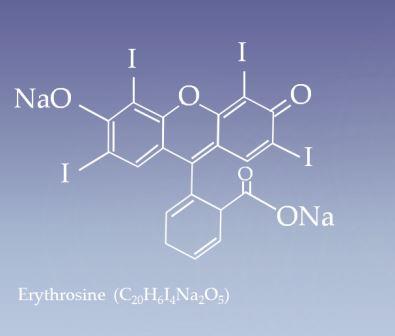
- 447 reads